Introduction
Hypokalemic periodic paralysis (HPP) is a neuromuscular disorder characterized by episodic painless severe muscle weakness associated with low serum potassium. It is usually triggered by high carbohydrate diet, trauma, exposure to cold or strenuous exercise. Case reports additionally have described paralysis triggered by β2-adrenergic bronchodilators. (1)(2)
HPP can be inherited or acquired. Inherited cases are secondary to muscle ion channel mutations (calcium or sodium). Acquired cases can occur secondary to renal tubular acidosis, acute gastroenteritis, primary hyperaldosteronism, Gitelman/Bartter syndrome and hyperthyroidism. (3) The latest is also known as Thyrotoxic periodic paralysis (TPP). (1)(4) TPP is a neuroendocrine emergency and although rare overall, it accounts for a significant amount of hospital admissions related to hypokalemic paralysis. As such, it should be in the differential diagnosis of weakness in a patient presenting with hyperthyroidism. (5)
Methodology
To perform this work, a MEDLINE search was conducted for articles published in the last 20 years using the mesh term “Hypokalemic periodic paralysis” and “Thyrotoxic periodic paralysis”. A total of 79 primary articles were reviewed. More relevant articles (n= 34) were selected.
Epidemiology
TPP is rare disorder with an incidence of approximately 2% in patients with thyrotoxicosis of any cause. (4) Most cases are sporadic, non-familial (6). This disorder has a higher expression in male due to low penetrance of the disease in females. When present in women, it is generally less severe. (7) Usually affects people in the range of 20 - 30 years of age with a male to female ratio of 20:1. (8)
TPP is more prevalent among Asian descents, incidence of TPP in Chinese and Japanese thyrotoxic patients has been reported at 1.8% and 1.9% respectively. (9) TPP has also been reported in young Hispanics, white and native American but actual incidence and prevalence rates are unknown as the literature consists of only case series and case reports. (10)(11) TPP should not be excluded from the differential diagnosis of paralysis solely because of the ethnicity of the patient. (12)
Pathogenesis
It is thought that metabolic abnormalities produced by hyperthyroidism in some way affect the permeability of the muscle cell membrane resulting in an influx of potassium. Proposed pathophysiology includes direct effects of thyroid hormone stimulation on the Na+-K+ ATPase activity (Figure 1) or indirect effect of thyroid hormone on insulin hypersecretion that will lead to intracellular potassium shift. (13)
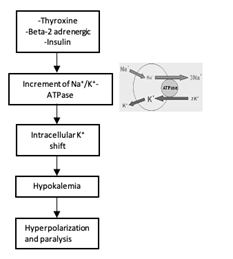
Figure 1 Mechanism of Thyrotoxic Periodic Paralysis (TPP). Thyroid hormone, insulin and B-2 adrenergic increase activity of Na⁺/K⁺-ATPase which leads to reduced K+ channel efflux and therefore hypokalemia. Hypokalemia produces hyperpolarization which leads to paralysis.
Thyroid hormone also increases the sensitivity of beta receptors, so catecholamine-mediated cellular potassium uptake is raised. This mechanism would account for reports that epinephrine worsens paralytic attacks, while propranolol has protective effects. (12)(13). This however will not explain why not all patients with hyperthyroidism or severe thyrotoxicosis develop TPP.
Several researchers have been trying to find out what makes some patients more predisposed to developing this type of paralysis. Some case series have identified a mutation with loss of function in the skeletal muscle specific inward rectifying potassium (Kir 2.6) channels, coded by KCNJ18 in approximately one-third of the patients with TPP, though others suggest these mutations are seldom pathogenic (14). Additional factors such as decreased potassium efflux must also play a role to cause clinically significant hypokalemia. Outward Kir current is decreased in intercostals muscle fibers of both patients with TPP and familial HPP. Moreover, insulin and catecholamine not only activate Na+-K+ ATPase but also inhibit Kir channels. Ryan DP and his colleagues in 2010 found that the defects of Kir2.6 were responsible for partial TPP in Brazilian, Singaporeans and Hong Kong patients. (13)(14)(15)(16)(17)
Graves’ disease is the most common etiology however TPP has also been reported in patients taking exogenous thyroid supplementation such as weight loss products and herbal medicines containing triiodothyronine (T3) and from thyrotropin-secreting pituitary adenomas (9)(11)(18)(19)
Clinical presentation
Hypokalemic paralysis, commonly observed in patients presenting to the emergency department, may be caused by neurologic, metabolic, or renal disorders (11).
In the case of TPP the classic triad of flaccid paralysis, signs of thyrotoxicosis and hypokalemia during a paretic crisis is the cornerstone for diagnosis. Degree of thyrotoxicosis can be mild and attacks occur suddenly and range from mild generalized weakness to flaccid paralysis (20)(21). Consciousness and respiratory muscles are preserved. Although few case reports have described fatal TPP due to dysrhythmias or respiratory paralysis (5)(20)(22). Frequency and intensity of attacks vary (from weeks to months and can occur every few weeks/months and can last from couple hours to days). Most commonly few hours to 2 days. (8)
There is a reported seasonal incidence, with increased frequency during summer months when temperature is around 30 °C (5). Typical presentation consists of proximal muscle weakness, with legs affected more than arms. Symmetrical involvement is more common with extensor muscles more involved than flexors, however it is not the rule. On physical exam: hyporeflexia or areflexia is noted with preservation of sensory modalities. It is the depressed deep tendon reflexes in a patient with hyperthyroidism (who usually has hyperreflexia) what should catch the attention of the physician evaluating the patient. (12) (Table 1)
Table 1 Clinical features of Thyrotoxic Periodic Paralysis (TPP)
| Clinical features |
| Age: Young (usually age 20-30 years of age) |
| Sex distribution: Predominantly male |
| Race: Asian, American Indian and Hispanic |
| Non hereditary, sporadic |
| Precipitated by high carbohydrate diet, trauma, exposure to cold or strenuous exercise |
| Recurrent episodes of acute paralysis with complete recovery |
| Proximal lower extremities affected > upper extremities and trunk |
| Clinically hyperthyroid |
| Hypokalemia |
| Electromyography (EMG): low-amplitude compound muscle action potential with no change after epinephrine |
*Clinical Presentation and distinguishing features seen in TPP. (35)
Attacks sometimes can be preceded by unspecific prodromal symptoms such as headaches and aches, pain and paresthesia in the affected extremities although some patients may not have any preceding symptoms. Potassium concentration during this attacks can vary from 2.2-3.2 mEq/L. Rare cases with levels <2 mEq/L (17), acidosis or alkalosis. (23) Arrhythmias although not part of this disorder can occur in the setting of severe hypokalemia, with rare cases reported of atrioventricular block, ventricular tachycardia, ventricular fibrillation, Brugada pattern and even cardiac arrest. (10)(24)(25).
Associated features will be related to overactive thyroid such as weight loss, heat intolerance, tachycardia, irritability or altered mental status, fine tremors and hyperdefecation. It is important to note that attacks of muscle weakness occur during the state of hyperthyroidism and never when the thyroid function is normal. (4)
Other laboratory findings will include suppressed TSH (< 0.1 milli-International Units/L) with high free T4 and total T3. If Graves’ disease is the culprit, patients will have positive Thyroid-stimulating immunoglobulin (TSI).
Diagnosis
TPP is a diagnosed based on clinical features along with supportive laboratory results. The classic triad of flaccid paralysis, signs of thyrotoxicosis and hypokalemia is diagnostic during an attack. Apart from measuring serum potassium, thyroid function test should be included: TSH, fT4 and T3. Sometimes symptoms and signs of hyperthyroidism may not be obvious but it should always be considered because it can be treated successfully. (6)
Other testing includes: EKG that could show depression of the ST segment, decrease in the amplitude of the T wave, and an increase in the amplitude of U waves but the predominant finding is sinus tachycardia. Arrhythmias, such as atrial fibrillation, ventricular fibrillation, atrioventricular block, and asystole, are occasionally seen (25)(26)(27). Phosphorus, calcium and magnesium should also be measured. Genetic testing is not required for suspected acquired HPP. (1)
EMG is not required but if done during an attack will show decreased amplitude of the compound muscle action potential (CMAP), with reduced motor unit recruitment or electrical silence, increased insertional activity, increased polyphasic motor unit potentials and reduced muscle fiber conduction velocity. Muscle biopsy is not usually performed. (1)
Other tests, such as thyroid ultrasonography, thyroid scintigraphy and radioiodine uptake, might be necessary to better evaluate hyperthyroidism in regards to its etiology and treatment plan. (12)
Treatment
Acute management of TPP involves oral or IV administration of potassium chloride (~60-120 mEq) which will revert weakness episodes. Patients who received potassium supplementation recovered their muscle strength sooner compared with patients who received normal saline (6.3 vs 13.5 h) (28). Recovery usually takes minutes to hours. It is important to note that potassium should not be given in solutions that contain dextrose as it will stimulate the release of insulin which can worsen weakness due to influx of potassium. Moreover, excessive intravenous potassium replacement may result in rebound hyperkalemia (K > 5 mmol/L in 30% and K > 6 mmol/L in 10 %) during recovery. Oral potassium, not exceeding 100mgEq per day may be favored but potassium supplementation for prophylaxis has not been effective. (26)(27)
The importance of recognizing the association of HPP with hyperthyroidism is very important because with successful treatment of the hyperthyroidism the episodes of paralysis disappear.
Treatment of hyperthyroidism is the mainstay of therapy in TPP, which usually results in remission of muscle paralysis. Treatment in TPP does not defer from usual hyperthyroidism treatment which includes thionamides such as methimazole (20-60mg once daily), propylthiouracil (200-600 mg divided in 3 doses), radioactive iodine ablation or with the discontinuation of thyroid hormone supplements in patients with exogenous thyrotoxicosis. (12)(29)
Most literature recommends also adding propranolol as it not only improves thyrotoxic symptoms but also improves paralysis by inhibiting the intracellular shift of potassium induced by epinephrine (11). Propranolol has long been recognized as efficacious in preventing recurrent attacks of TPP and it reduces the incidence of spontaneous episodes (13). Selective β-blockers, including metoprolol, do not protect patients from paralytic attacks, which is consistent with the specific role of the β2 receptor in mediating the catecholamine-induced increase in Na+-K+-ATPase activity in skeletal muscle. (30)
Recovery is usually complete and between attacks patients are asymptomatic aside from hyperthyroid symptoms.
The definitive treatment of TPP is producing a euthyroid state, initially with antithyroid medications and subsequently with thyroidectomy or radioiodine thyroid ablation. Until euthyroid state is achieved, patients should continue propranolol and avoid precipitating factors. (10)(31)(32) Once euthyroidism has been established, the patients no longer present with paralytic attacks and show good clinical outcomes if treatment compliance is achieved. (11)(33)(34)